Hydrometallurgy – the treatment of primary raw materials (minerals or mineral concentrates) or secondary raw materials (end-of-life products) by aqueous solutions, in order to achieve the dissolution of the metals to be recovered.
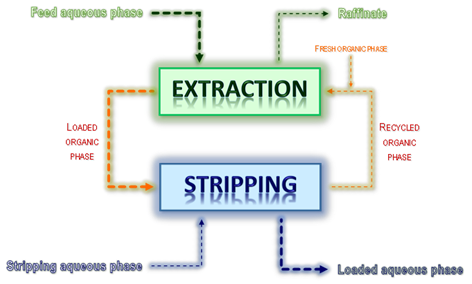
Solvent extraction in hydrometallurgy – a purification or concentration unit operation. The feed aqueous solution to purify or concentrate is put in contact with a suitable organic solvent – EXTRACTION step. Afterwards, the loaded organic phase is again contacted with an adequate aqueous solution to revert the equilibrium, the metal ion being then transferred to a new aqueous phase – STRIPPING step. The success of the process is guaranteed by the regeneration of the organic solution after stripping of the metal ion – allowing its recycling – and if the organic solvent has been efficient and selective towards the metal species to be recovered (see a simplified scheme below).
Extractant – one or more active ingredients which is (are) responsible for the extraction of the desired metal species.
Diluent – the “support” of the organic solvent, ideally inert during extraction, used to improve the physical properties (density, viscosity, etc.) or the extractive properties (e.g., selectivity) of the extractant.
Modifier – substance added to the organic solvent, usually to improve the solubility of the extractant (and extracted metal species) in the solvent. It can also be used to break stable emulsions.
Distribution ratio – total analytical concentration of a metal in the organic phase to its total analytical concentration in the aqueous phase after extraction, usually determined at equilibrium.
Stripping agent – water, or aqueous solutions of acids, bases or salts, used to transfer the desired metal ions from the loaded organic solvent to a new aqueous solution.
To acquire more information about solvent extraction and other hydrometallurgical unit operations, readers can follow, for example, the links indicated below,
- http://en.wikipedia.org/wiki/Hydrometallurgy
- http://www.sciencedirect.com/science/article/pii/S1007021406701687
or consult the following books:
- M.L. Free, Hydrometallurgy: Fundamentals and Applications, The Minerals, Metals & Materials Society, John Wiley & Sons, Inc. New York, 2013, 444 pp.
- M. Cox, Solvent Extraction in Hydrometallurgy, in Principles and Applications of Solvent Extraction, 2nd Ed., J. Rydberg, C. Musikas, G.R. Choppin, Eds., Marcel Dekker Inc., New York, 2004, pp. 455-505.



